Description
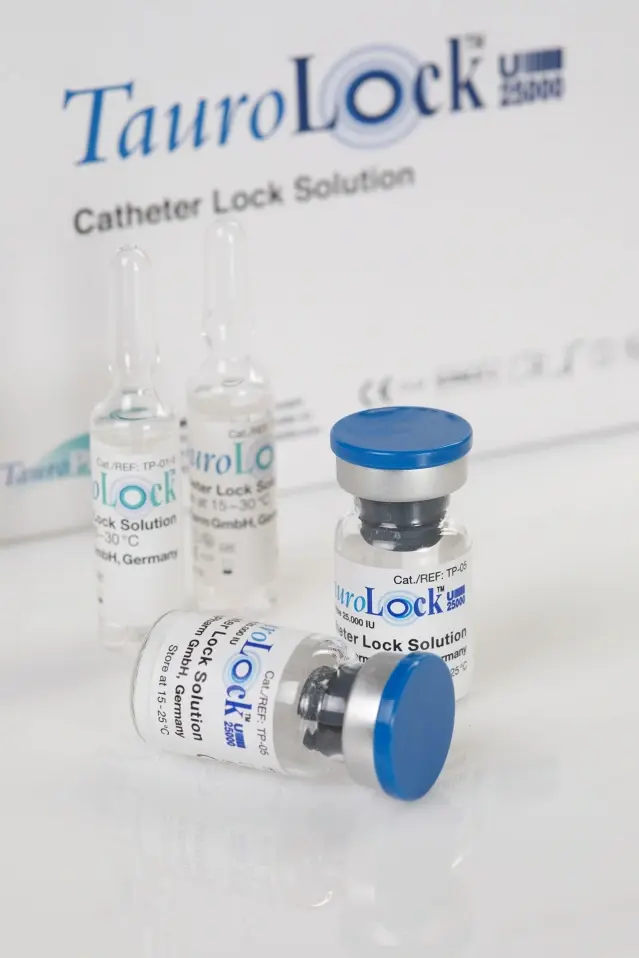
When it comes to ensuring the longevity and functionality of catheters, a taurolock catheter lock solution emerges as a vital element. TauroLock U25000 Vial steps up as an advanced solution, revolutionizing catheter care. In the ever-evolving landscape of medical technology, the demand for effective catheter maintenance has surged, making innovative options like TauroLock U25000 Vial a necessity. Explore the ease of acquiring TauroLock U25000 Vial online in Pakistan.
TauroLock U25000 Vial is meticulously designed to sustain catheter patency and minimize the risk of catheter-related infections. Its specialized formulation acts as a protective barrier, thwarting clot formation and microbial colonization, thus extending the catheter’s useful life. Whether you’re a dedicated healthcare professional or an individual managing a catheter at home, TauroLock U25000 Vial ensures optimal catheter upkeep.
The growing need for top-tier healthcare products has underscored the importance of online accessibility. Now, procuring TauroLock U25000 Vial online in Pakistan is an effortless endeavor. With a few clicks, you can contribute to the well-being of catheter-dependent individuals, enhancing their quality of life.
At Algene Medical, our commitment to delivering impactful healthcare solutions remains steadfast. TauroLock U25000 Vial stands as a testament to our unwavering dedication to medical innovation and excellence. With patients and healthcare providers at the core of our mission, we proudly introduce TauroLock U25000 Vial as the ultimate catheter lock solution.
In conclusion, TauroLock U25000 Vial embodies catheter care evolution. Its capacity to prevent clot formation and microbial colonization positions it as an essential component of catheter maintenance. Embrace the convenience of purchasing TauroLock U25000 Vial online in Pakistan, joining us in promoting superior healthcare for catheter-dependent individuals. With TauroLock U25000 Vial, your catheter care journey reaches new heights.
TauroLock™-U25.000 provides a particularly strong prophylaxis against occlusion, as urokinase serves to break up blood clotting.
TauroLock™-U25.000 is based on three active ingredients:
- taurolidine
- citrate (4 %)
- urokinase (25,000 units)
Taurolidine has a broad antimicrobial activity (incl. MRSA and VRE), while citrate works as an anticoagulant. At the same time, urokinase yields a thrombolytic effect.
Contraindications
TauroLock™-U25.000 is contraindicated:
– for patients with a known allergy to (cyclo)-taurolidine, citrate or urokinase,
– or when a patient is currently taking medication with known adverse interaction to citrate, (cyclo)-taurolidine or urokinase.
Safety
Prior to the next treatment, TauroLock™-U25.000 must be aspirated and discarded according to the institution’s policy for infectious waste disposal.
Package Sizes
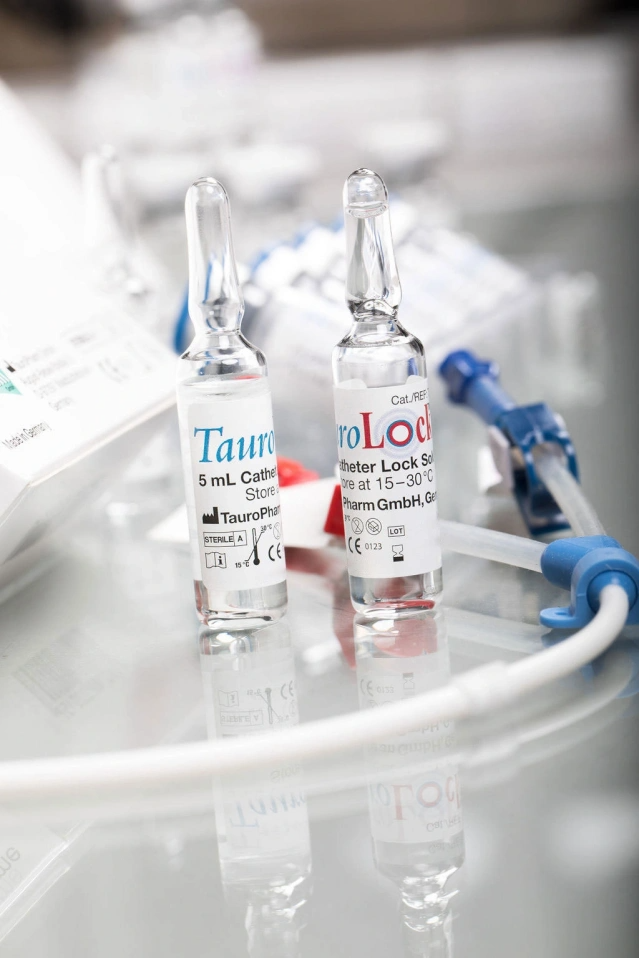
You may purchase TauroLock™-U25.000 in vials of 5 ml (single dose, 5 vials per box).
Storage & Transport
TauroLock™-U25.000 must be stored at 15 °C to 25 °C.
Please note that you must not freeze TauroLock™-U25.000 under any circumstances.
Jasmine Block, Bahria Town
Lahore 53720, Pakistan
Faq's
NutriLock™ and TauroLock™ are catheter lock solutions for tunneled and non-tunneled vascular access and port systems for the prevention of catheter-associated infections and catheter flow problems.
NutriLock™ contains taurolidine as an antimicrobial ingredient. In addition to taurolidine, TauroLock™ solutions also contain 4 % citrate for the maintenance of catheter patency.
In addition to taurolidine and 4 % citrate, TauroLock™-HEP100 contains additional 100 IU/ml heparin to improve patency. TauroLock™-HEP500 contains additional 500 IU/mL heparin.
Тhe most effective prophylaxis against catheter occlusion is maintained with the regular use of TauroLock™-U25.000, containing 25.000 IU urokinase (5.000 IU/mL), taurolidine and 4 % citrate. This reduces flow problems in the catheter substantially (see lock solutions in dialysis).
The decision as to which catheter lock solution is most advantageous depends on the patient’s individual situation. Alternating use (e.g. TauroLock™-HEP500, TauroLock™-U25.000) is feasible.
Heparin and 4 % citrate only have anticoagulant effects, but no bactericidal properties. Consequently, contamination of the catheter can lead to bacteraemia.
TauroLock™ solutions are used to prevent both infections and occlusions in catheter and port systems. NutriLock™ acts prophylactically against catheter infections.
Prophylactic use of TauroLock™ solutions prevents the formation of biofilm and thus maintains the flow rate in the catheter and/or port. The fibrinolytic activity of TauroLock™-U25.000 further improves patency by dissolving already formed clots.
Due to the citrate content (citrate content: 4 %), overly rapid application into the bloodstream can lead to mild hypocalcaemic effects (e.g. metallic taste).
A citrate concentration of 4 % is recommended by US regulatory authorities and others.
- In the context of one case of fatality, the FDA issued an advisory not to use a product with a higher citrate content (Tricitrasol, 46.7 %). Lock solutions of this type had to be recalled from the US market. Their use is no longer approved.
- Likewise, over-instillation of less than 1 ml per lumen of a 30% citrate solution induced transient cardiac arrest in two cases in the Netherlands (Punt CD, Boer WE, Cardiac arrest following injection of concentrated trisodium citrate, Clinical Nephrology 2008, 69 (4), 317-318).
- A high citrate content (46.7%) can also lead to embolic events which might be triggered by protein precipitation (Davenport A , Willicombe MK, Vernon K, Embolic complications from central venous hemodialysis catheters used with hypertonic citrate solution, American Journal of Kidney Diseases, 2010, 55, 348-351 / Schilcher G, Scharnagl H, Horina JH, Ribitsch W, Rosenkranz AR, Stojakovic T and Polaschegg H-D, Trisodium citrate induced protein precipitation in haemodialysis catheters might cause pulmonary embolism, Nephrol Dial Transplant (2012) 0: 1–5).
Instructions and guidelines
- Before instilling the lock solution, flush the vascular access system with at least 10 ml of physiological saline with the pulsatile flushing technique.
- Use a suitable syringe to remove NutriLock™ or a TauroLock™ solution from the container.
- Instill the lock solution into the access device in a quantity sufficient to fill the lumen completely. The specific filling volume is stated in the instructions for use of the access system.
- NutriLock™ or TauroLock™ solutions remain inside the access system until the next treatment.
- Before starting the next treatment, the lock solution must be aspirated.
- Flush the vascular access system with 10 ml of physiological saline.



Reviews
There are no reviews yet.